 Fractional crystallisation can be used to obtain pure para product, relying on the principle that it is less soluble than the ortho and thus will crystallise first.Column chromatography will often separate these isomers, as the ortho is more polar than the para in general.Several methods exist in order to separate these isomers: Separation of ortho and para isomers īecause electron donating groups are both ortho and para directors, separation of these isomers is a common problem in synthetic chemistry. However, the para isomer usually has the highest melting point, and the lowest solubility in a given solvent, of the three isomers. The toluidines serve as an example for these three types of substitution.Įlectron donating groups, for example amino, hydroxyl, alkyl, and phenyl groups tend to be ortho/ para-directors, and electron withdrawing groups such as nitro, nitrile, and ketone groups, tend to be meta-directors.Īlthough the specifics vary depending on the compound, in simple disubstituted arenes, the three isomers tend to have rather similar boiling points. In para-substitution, the substituents occupy the opposite ends (positions 1 and 4, corresponding to R and para in the diagram).In meta-substitution the substituents occupy positions 1 and 3 (corresponding to R and meta in the diagram). 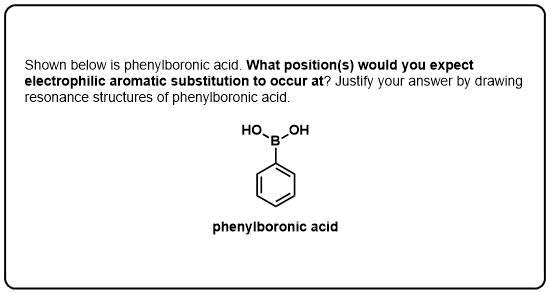 
In the diagram, these positions are marked R and ortho. In ortho-substitution, two substituents occupy positions next to each other, which may be numbered 1 and 2.See also: Electrophilic aromatic substitution 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
